THE PROBLEM
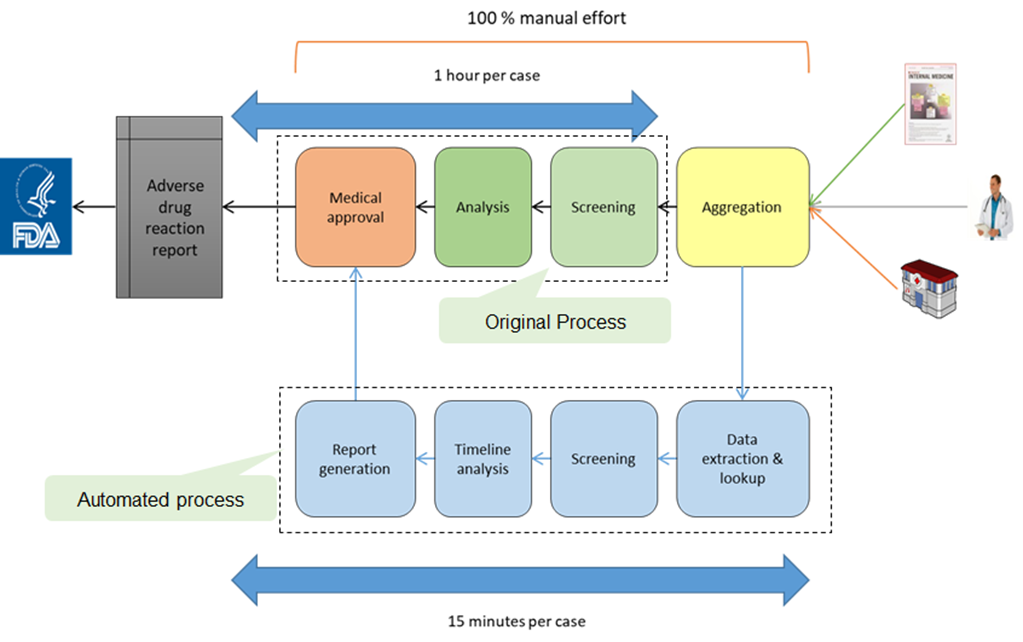
In the pharmaceutical industry, effect of a drug on patients (adverse symptom, conflicts etc.) is tracked post FDA approval and market release. To achieve this, a large number of patients who have been prescribed the drug are tracked and constantly monitored for: adverse symptoms, conflict with ongoing prescribed drugs etc. Tracking patients relies on multiple documents with unstructured data being collated (prescriptions, case histories, patient discharge forms etc.). Hence the case preparation process was completely manual and consequently costly, time consuming and error prone. Client, a global consulting firm, which was involved in the case preparation process wanted to automate the process.

INXITE OUT APPROACH
Natural Language Processing techniques were utilized to extract information from multiple structured and unstructured documents and aggregated in case file. Custom text processing techniques were used to parse and understand the structure of the documents and to consequently tokenize the text for further processing. Medical and Pharma dictionaries like MedDRA, WHO-DD, RxNorm, SNOMED-CT for Medical Coding & Disambiguation were further used to interpret the tokens in a medical context and to cluster the interpreted entities into relevant case files.
RESULT
Successfully automated the case preparation process reducing processing time by ~75%








